Rally #188-1-2377
Quidel #20377
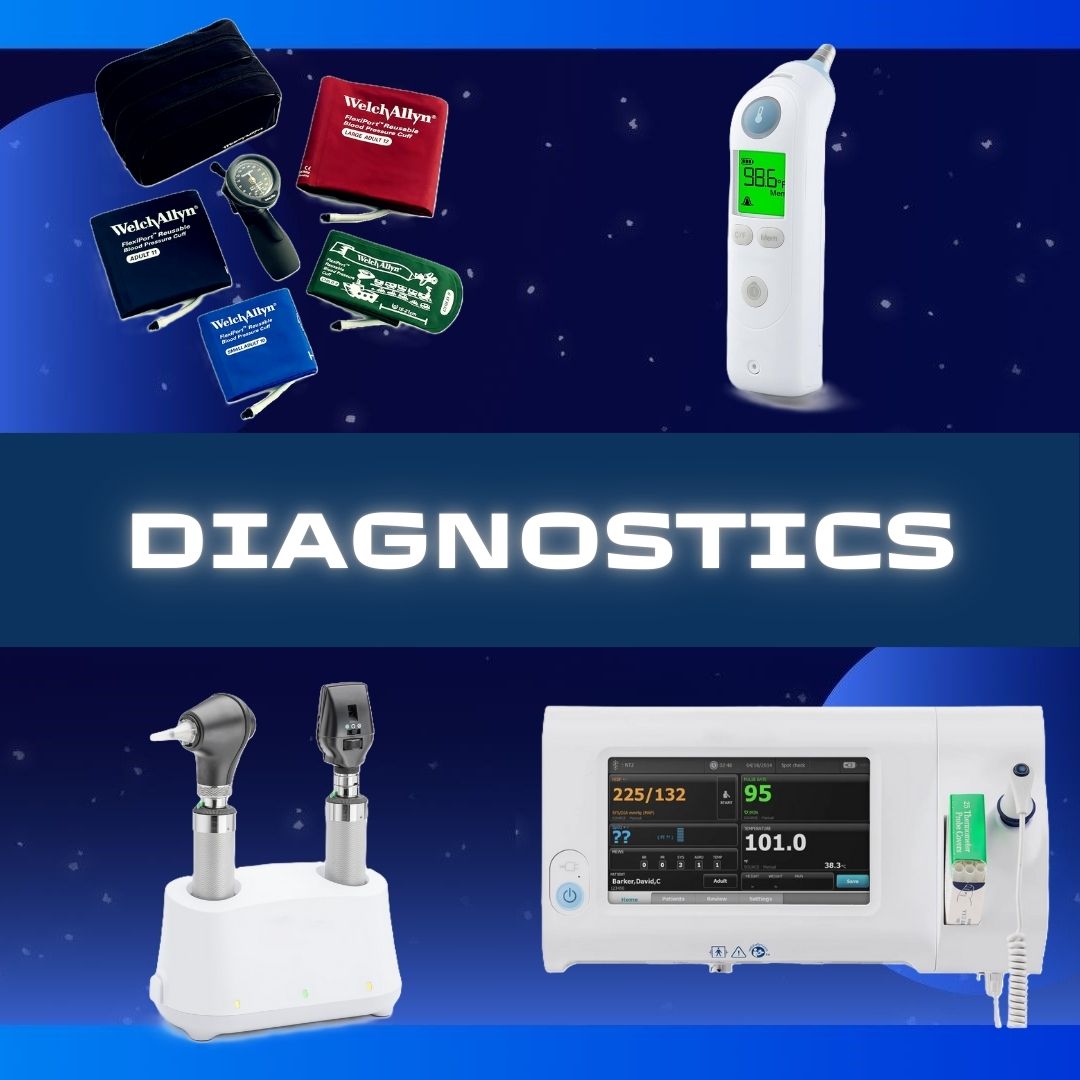
Sofia® 2 Flu + SARS Antigen FIA (25 Test/KT 12 KT/CS)
Respiratory Test Kit, SARS-CoV-2 / Flu A+B, Lateral Flow Immunofluorescent Sandwich Assay, Nasal Swab, Rx Only, CLIA Complexity Waived
Features
- One sample, three results mean time savings, material conservation, and patient comfort
- Rapid results in 15 minutes to support efficient dispositioning of patients
- Objective, accurate, reliable results without cross-reactivity to seasonal coronaviruses
- Room temperature storage
This test is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation.
The test does not differentiate between SARS-CoV and SARS-CoV-2 viruses and is not intended to detect influenza C antigens.
All negative results are presumptive and should be confirmed with a FDA-cleared molecular assay. Negative results do not rule out SARS-CoV-2 or influenza virus infection and should not be used as the sole basis for treatment or patient management decisions.
The Sofia 2 Flu + SARS Antigen FIA is intended for use on the Sofia 2 only and by medical professionals or trained operators who are proficient in performing tests using the Sofia 2 Instrument.
Important Note:
- Product is non-returnable; please make sure it is the right product before ordering.
- If put into refrigeration, do not freeze.
- For in vitro diagnostic use.
- Rx Only.
- For use under an Emergency Use Authorization (EUA) only.
- For more information on this product's EUA, please go to the EUA section below.
Warning: This product can expose you to chemicals known to the State of California to cause cancer. For more information, go to www.P65Warnings.ca.gov.
| Rally # | 188-1-2377 |
| Manufacturer # | 20377 |
| Brand | Sofia® 2 |
| Manufacturer | Quidel |
| Country of Origin | United States |
| Application | Respiratory Test Kit |
| CLIA Complexity | CLIA Waived |
| For Use With | For use with Sofia 2 Fluorescent Immunoassay Analyzer |
| Number of Tests | 25 Tests |
| Reading Type | Machine Read |
| Sample Type | Nasal Swab Sample |
| Specialty | Immunoassay |
| Test Format | Cassette Format |
| Test Kit Type | Rapid |
| Test Name | Flu + SARS Antigen FIA |
| Test Type | Fluorescence Immunoassay (FIA) |
| Time to Results | 15 Minute Results |
| UNSPSC Code | 41116144 |
| Kit Storage Conditions | Room Temperature (15 °C to 35 °C / 59 °F to 86 °F) |
| Type | PPA (Sensitivity) | NPA (Specificity) |
| Influenza A | NS - 90%, NPS - 97.1% | NS - 95%, NPS - 94.6% |
| Influenza B | NS - 89%, NPS - 90% | NS - 96%, NPS - 97% |
| SARS-CoV-2 | 95.2% | 100% |
Configurations
| 20377 | Sofia® 2 Flu + SARS Antigen FIA Test Kit |
| 20391 | Sofia 2 Flu + SARS Antigen FIA Control Set |
Reagents and Materials Supplied - 20377 (25-Test Kit)
- Individually Packaged Test Cassettes (25): Mouse monoclonal anti-influenza A and anti-influenza B antibodies; Monoclonal anti-SARS antibodies
- Reagent Tubes (25): Lyophilized buffer with detergents and reducing agents
- Reagent Solution (25): Ampoules with salt solution
- Sterile Nasal Swabs (25)
- Small, Clear 120 µL Fixed Volume Pipettes (25)
- Flu + SARS Positive Control Swab (1): Swab is coated with non-infectious recombinant influenza A, influenza B, and SARS antigens
- Negative Control Swab (1): Swab is coated with heat-inactivated, non-infectious Streptococcus C antigen
- Package Insert (1)
- Quick Reference Instructions (1)
- QC Card (located on kit box)
Manufacturer Documentations
- Sofia 2 Flu + SARS Antigen FDA EUA Letter
- SARS-CoV-2 Variant Detection Bulletin
- Quidel 12.28.21 Omicron Variant Letter
- CLSI - Sofia 2 Flu + SARS Antigen FIA
- FAQ - Sofia 2 Flu + SARS Antigen FIA
- HCP Fact Sheet - Sofia 2 Flu + SARS Antigen FIA
- Package Insert - Sofia 2 Flu + SARS Antigen FIA
- Package Insert - Sofia 2 Flu + SARS Antigen FIA Control Set
- Patient Fact Sheet - Sofia 2 Flu + SARS Antigen FIA
- Quick Reference Instructions - Sofia 2 Flu + SARS Antigen FIA
- Quiz - Sofia 2 Flu + SARS Antigen FIA
- SDS - Sofia 2 Flu + SARS Antigen FIA
- Sales Sheet - Sofia 2 Flu + SARS Antigen FIA
- Sample Collection - Sofia 2 Flu + SARS Antigen FIA
- Technical Bulletin - Firmware 1.12 Update
- Technical Bulletin - Sofia 2 Flu + SARS Antigen FIA (Liquid Proficiency Sample Testing Procedure)
- Training Tools - Sofia 2 Flu + SARS Antigen FIA
Promotions are currently not available at this time. Please check back later.
- Reimbursements Information -
This product maybe reimbursable, depending on various factors including current law and policies. Rally Inc. does not handle these reimbursements; instead, each reimbursement is handled solely between you, the customer, and the manufacturer, QuidelOrtho. Rally Inc. does not oversee these reimbursements. To check eligibility and get your reimbursements, please follow the manufacturer's reimbursements instructions.You can find more informations on reimbursement details here.
EUA - Emergency Use Authorization
In the USA, this product has not been FDA cleared or approved, but has been authorized by FDA under an Emergency Use Authorization. This product has been authorized only for the detection of proteins from SARS-CoV-2, influenza A, and influenza B, not for any other viruses or pathogens. The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug, and Cosmetic Act, 21 U.S.C. §360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.The test has been authorized for emergency use by FDA under an Emergency Use Authorization (EUA) for use by authorized laboratories certified under the CLIA that meet the requirements to perform moderate, high or waived complexity tests. This test is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation.